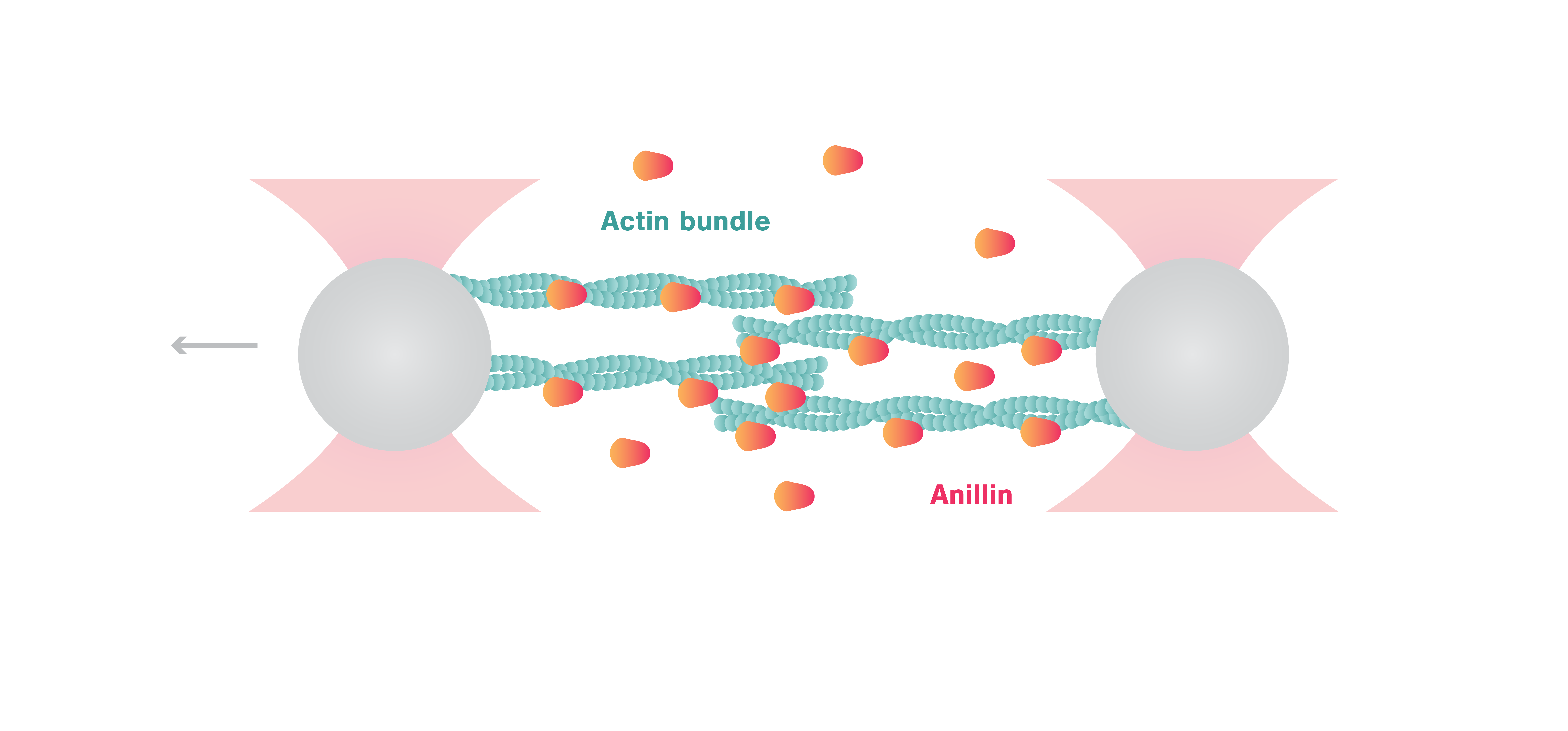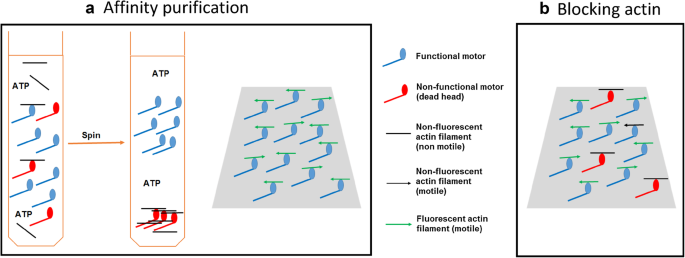
Comparative analysis of widely used methods to remove nonfunctional myosin heads for the in vitro motility assay | SpringerLink
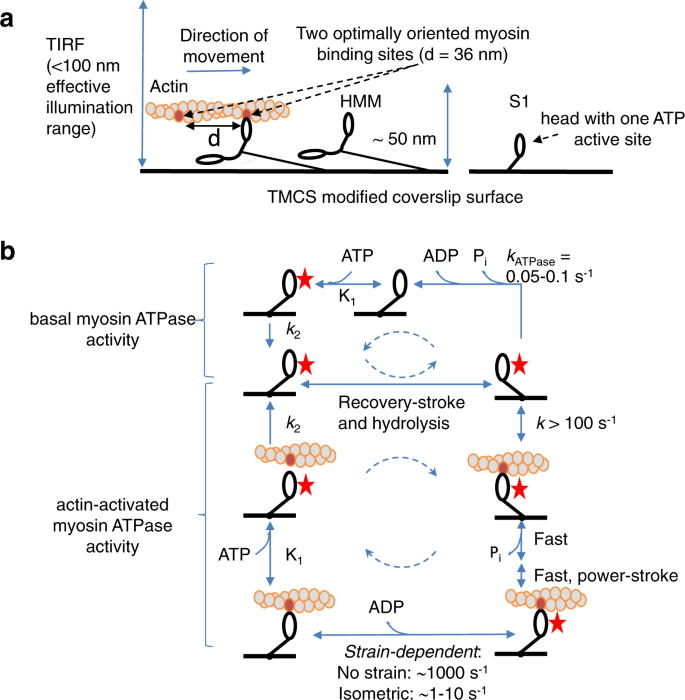
Single molecule turnover of fluorescent ATP by myosin and actomyosin unveil elusive enzymatic mechanisms | Communications Biology

Twirling of Actin by Myosins II and V Observed via Polarized TIRF in a Modified Gliding Assay: Biophysical Journal

Twirling Motion of Actin Filaments in Gliding Assays with Nonprocessive Myosin Motors: Biophysical Journal

Velocities of unloaded muscle filaments are not limited by drag forces imposed by myosin cross-bridges | PNAS

A mixed-kinetic model describes unloaded velocities of smooth, skeletal, and cardiac muscle myosin filaments in vitro | Science Advances

Actin-binding domain of Rng2 sparsely bound on F-actin strongly inhibits actin movement on myosin II | Life Science Alliance
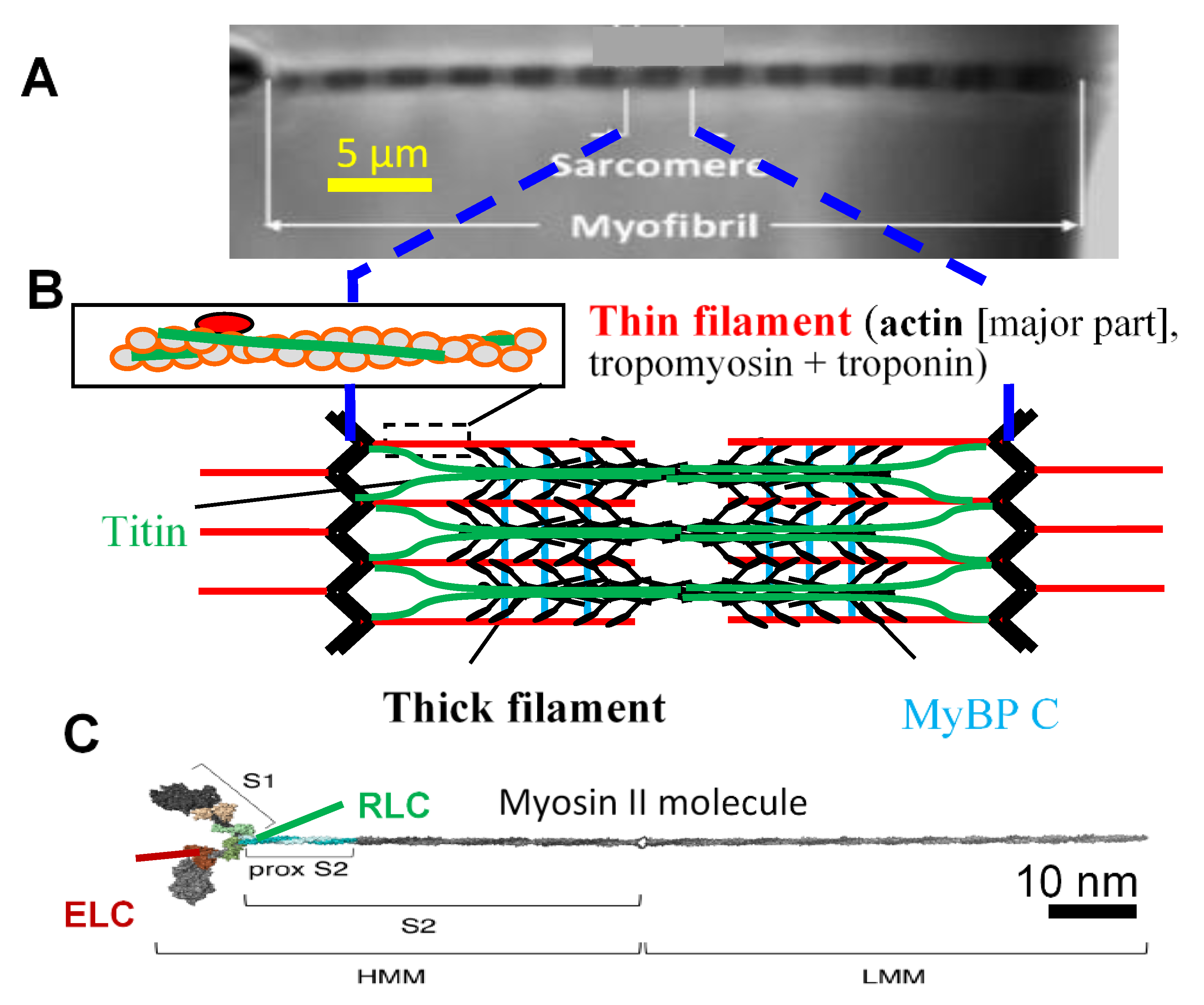


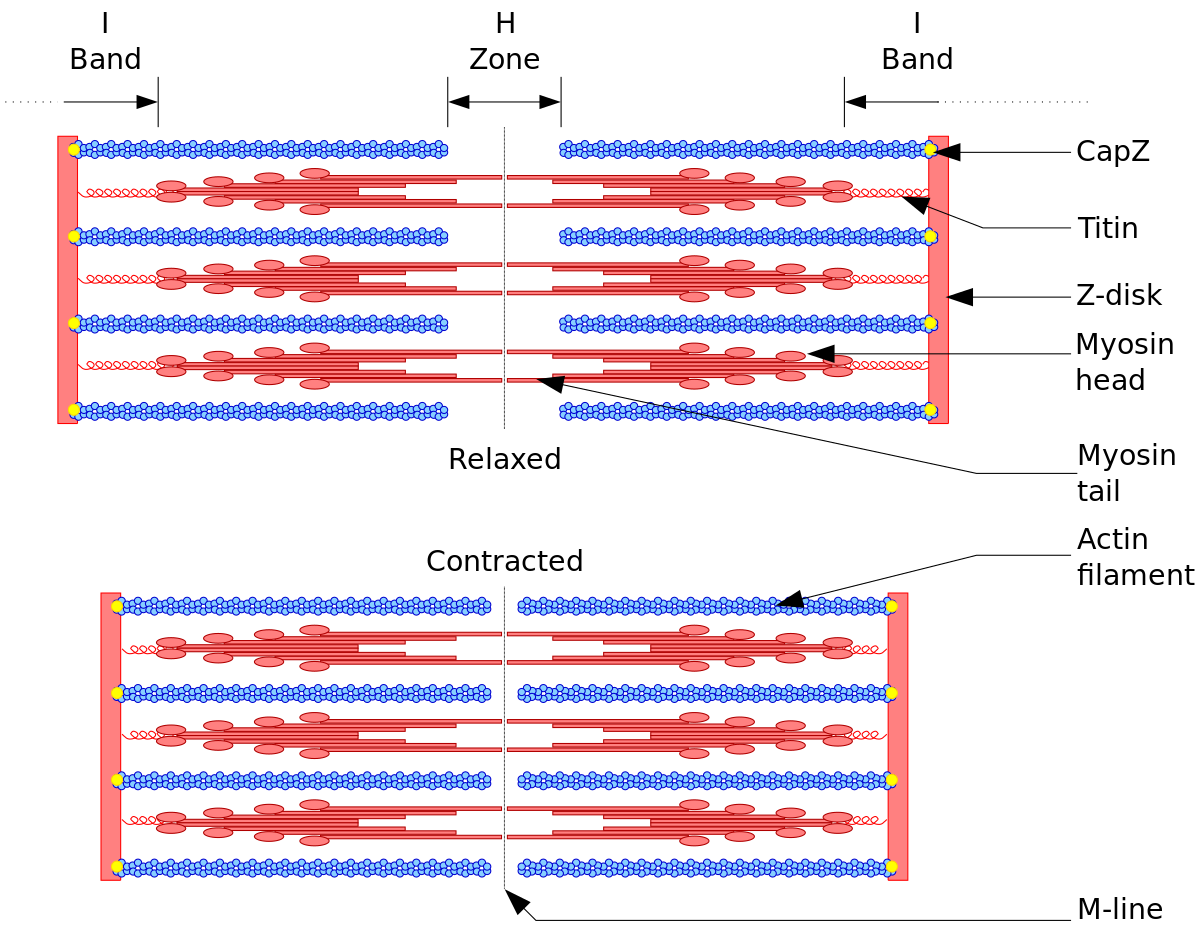


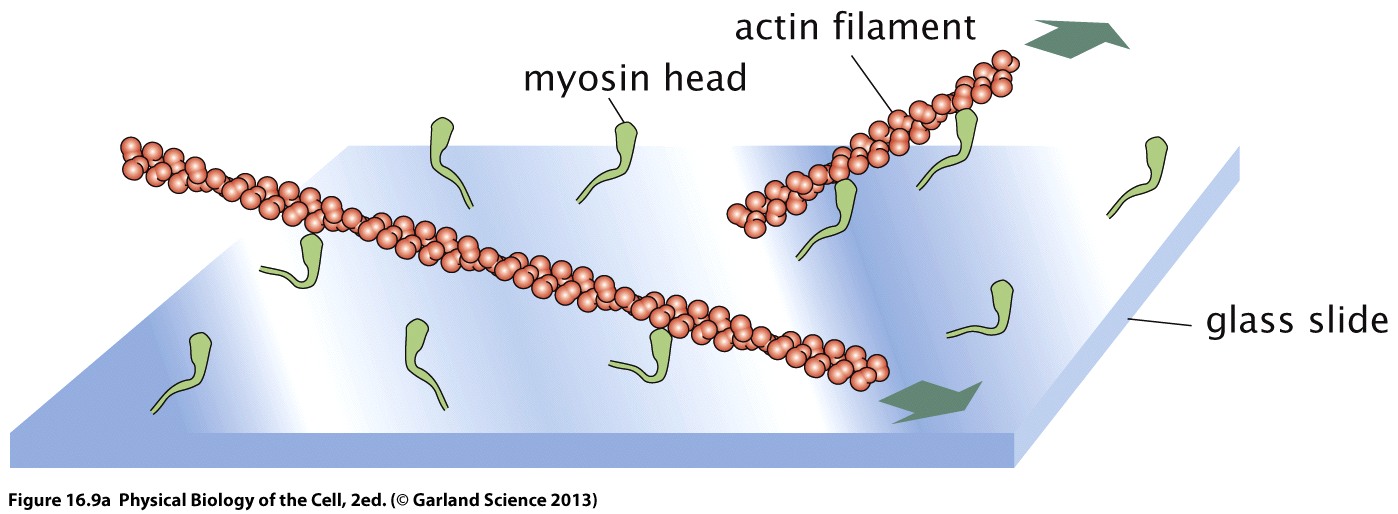

![PDF] MICROFILAMENTS AND INTERMEDIATE FILAMENTS | Semantic Scholar PDF] MICROFILAMENTS AND INTERMEDIATE FILAMENTS | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bdf0afd51b15ec4da9ef8cfd50bb2209700b3fb7/15-Figure3-24-1.png)